The first local operational document implementing Order No. 818 has been released! The "Guangdong Province Biomedical New Technology (Stem/Somatic Cell Field) Clinical Research Registration Work Guide
2026/02/14


















Cell therapy product safety evaluation platform
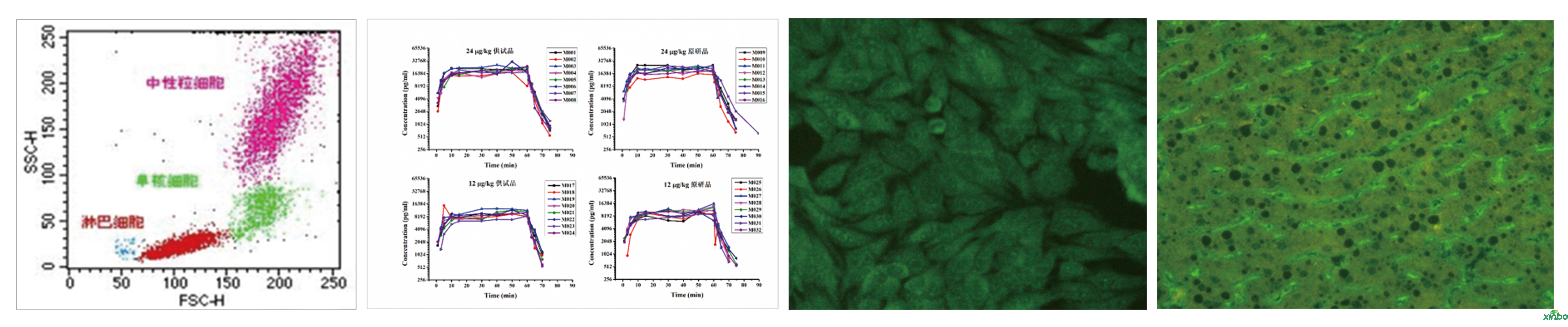
Shandong Xinbo is capable of conducting safety evaluations for cell therapy products. Currently, we have conducted pharmacodynamic studies, pharmacokinetic studies, and a full set of preclinical safety evaluations for CGT products such as NK, MSC, and CAR-T.
■ Cell counting
■ Pharmacodynamic study
■ Single-dose toxicity test (rodents)
■ Repeated-dose toxicity test (rodents)
■ Safety pharmacology test
■ Pharmacokinetic test (including method establishment and validation)
■ Local toxicity test
■ Immunogenicity test (including methodological establishment and validation)
■ Tumorigenicity test
■ Tumorigenicity test
■ Soft agar cloning assay
About Shandong Xinbo
Shandong Xinbo, established in April 2010, boasts laboratories in Jinan and Linyi. It is recognized as a national high-tech enterprise, a Shandong enterprise technology center, and a Shandong gazelle enterprise. As a research service enterprise, it integrates pharmacodynamic research, toxicological research, drug analysis and testing services, animal pathology inspection, and medical device evaluation services.
Shandong Xinbo has completed multiple preclinical safety studies for new drugs in the CGT field, successfully assisting in obtaining IND approvals for various drugs. Meanwhile, in the research and development of new biomedical technologies and clinical research filing work, the company has completed and is currently conducting multiple preclinical studies, providing solid technical support for clinical research filing.
Tel: 0534-5056588 0531-68801033
