Shandong Xinbo helps Class 1.1 pediatric fever patch get approved for clinical trials
2026/03/27
In the innovative research and development process of "Mugui Antipyretic Patch", Shandong Xinbo relies on its core technology platform - the pediatric drug safety evaluation platform, focusing on the research and development characteristics of traditional Chinese medicine external patches and the rigid demand for pediatric drug safety, to provide full-process and standardized preclinical safety evaluation technical services for this product.
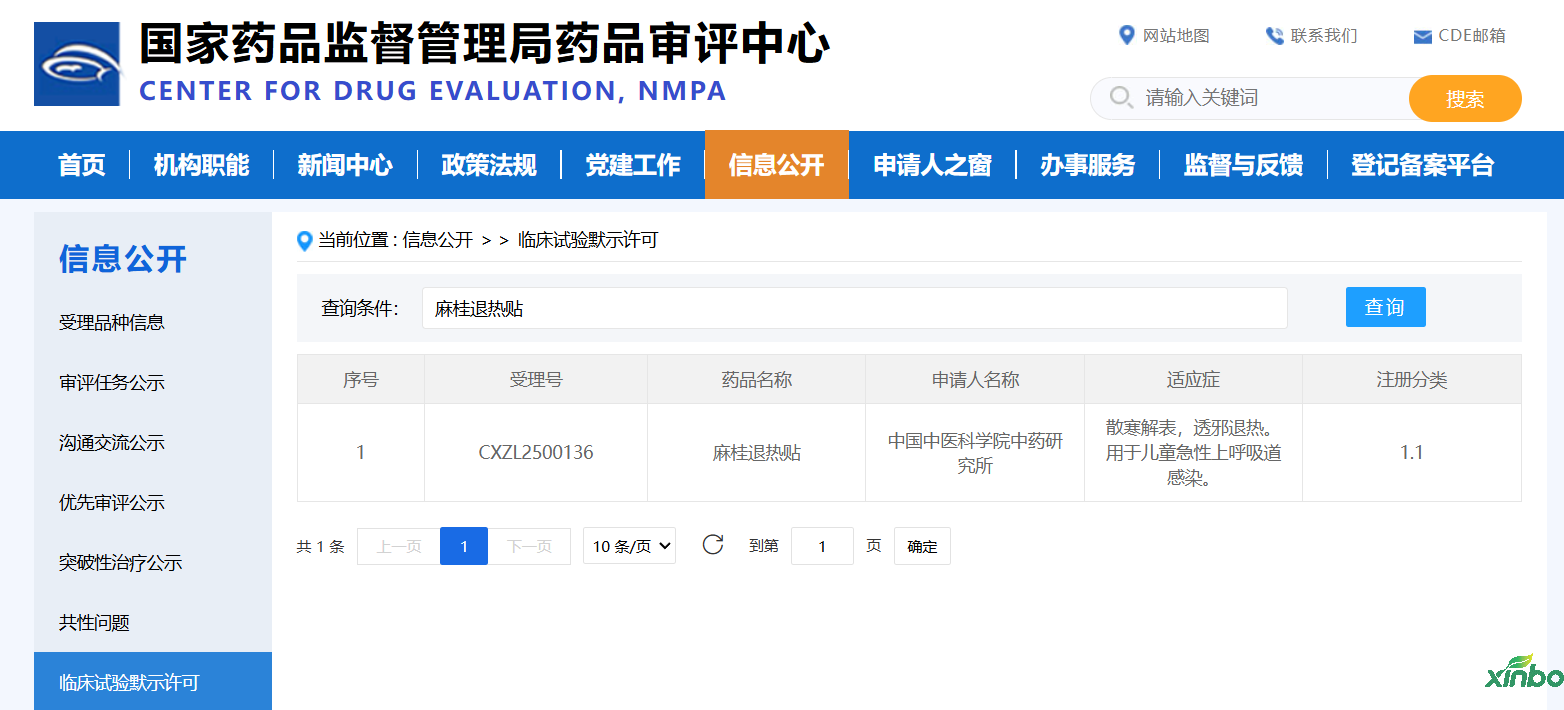
"Function and efficacy of 'Mugui fever-reducing patch'"
"Mugui Antipyretic Patch" serves as a typical representative of external preparations, offering unique advantages in terms of administration method and safety properties compared to oral antipyretic drugs. Currently, the mainstream antipyretic drugs in the market are mostly oral preparations. Children often have low compliance due to the unpleasant taste, while the elderly are prone to adverse reactions due to gastrointestinal function. However, traditional Chinese medicine antipyretic gel patches, which work through external application, avoid gastrointestinal irritation, making them more convenient and safe to use. This aligns with consumers' demand for green and healthy therapies. Among them, antipyretic patches with pure Chinese medicine ingredients have gained increasing recognition due to their natural and mild properties, becoming an important direction for innovation and development in the antipyretic field.
Pediatric drug safety evaluation platform
Shandong Xinbo has established a mature pediatric drug safety evaluation platform and developed gavage administration techniques for rodents at 4 days old and non-rodents (dogs) at 7 days old, successfully completing safety evaluations for multiple pediatric drugs.
■ Single-dose toxicity test (rodents, non-rodents)
■ Repeated-dose toxicity tests (rodents, non-rodents)
■ Growth and development monitoring of young animals
■ Reflex development detection in young animals
■ Detection of behavioral indicators in young animals
■ Pharmacokinetic test
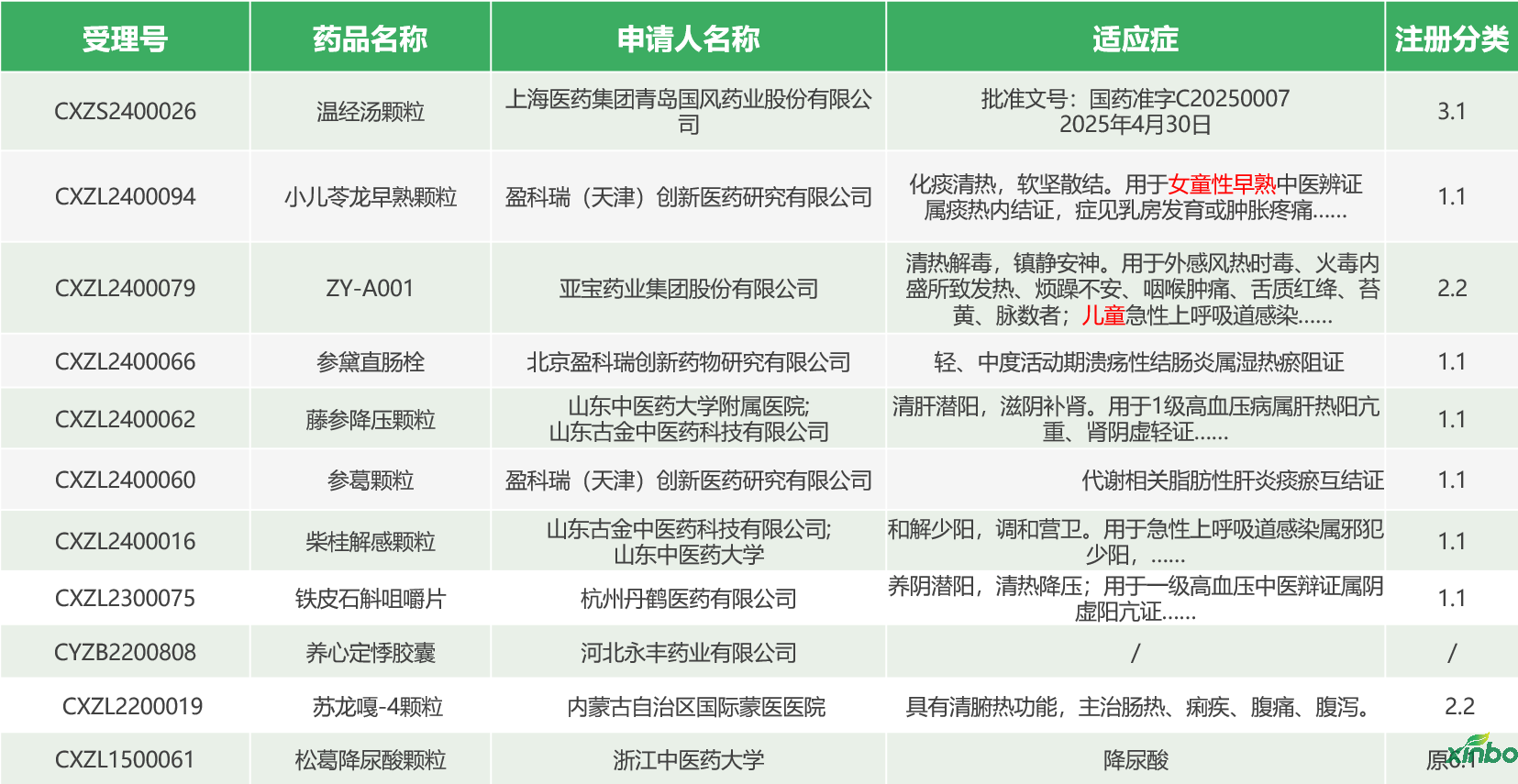
Shandong Xinbo has contributed to the approval of clinical Chinese medicine-related achievements
About Shandong Xinbo
Shandong Xinbo was established in April 2010 and has laboratories in Jinan and Linyi. It is a national high-tech enterprise, a Shandong enterprise technology center, and a Shandong gazelle enterprise. It is a research service enterprise integrating pharmacodynamic research, toxicological research, drug analysis and testing services, animal pathology testing, and medical device evaluation services. It has completed preclinical safety evaluations for over 400 innovative drugs, including chemical drugs, traditional Chinese medicine and natural products, biological products, preventive and therapeutic vaccines, monoclonal antibodies, cell therapy products, medical devices, and other varieties.
Tel: 0534-5056588, 0531-68801033
Website: www.xinbo.cn
Email: bd@xinbo.cn
Address: No. 1308, Fumin Road, Linyi Economic Development Zone, Dezhou City, Shandong Province (Linyi Laboratory)
5th Floor, Building A, Lishan International Cell Medicine Industrial Park, Jinan City, Shandong Province (Jinan Laboratory)

